
___________________________________________________________________________
2018/SCSC/WKSP8/009
Malaysia Maximum Residue Limit Regulatory
Enforcement
Submitted by: Malaysia
Workshop on a Trade Facilitative
Approach to Maximum Residue Limit
Compliance
Brisbane, Australia
9-10 October 2018

MALAYSIA MRL REGULATORY
ENFORCEMENT
1
Food Safety and Quality Programme
Ministry of Health Malaysia
APEC FSCF PTIN MRL Harmonization: A Trade Facilitative Approach to MRL Compliance
9-10 October 2018, in Brisbane, Australia
RAFEAH SIBIL
Senior Principal Assistant Director
Food Safety & Quality Division
Website : http://fsq.moh.gov.my

1
2
3
4
MRL
Domestic and Import
MRL in Regulations
MRL Domestic Monitoring
Clearance of MRL Import


OBJECTIVE
FOOD SAFETY AND QUALITY
PROGRAMME
4
General
Objective
To protect the public against
health hazards and fraud in the
preparation, sale and storage of
food.
To facilitate food trade
Food Safety and Quality Programme
Ministry of Health Malaysia

COMPETENT AUTHORITY
The Food Safety and Quality
Division, Ministry of Health is the
regulatory authority for food safety
in Malaysia
5
Food Safety and Quality Programme
Ministry of Health Malaysia
PROGRAMME STRUCTURE
Food Safety and Quality Programme
Ministry of Health Malaysia
6
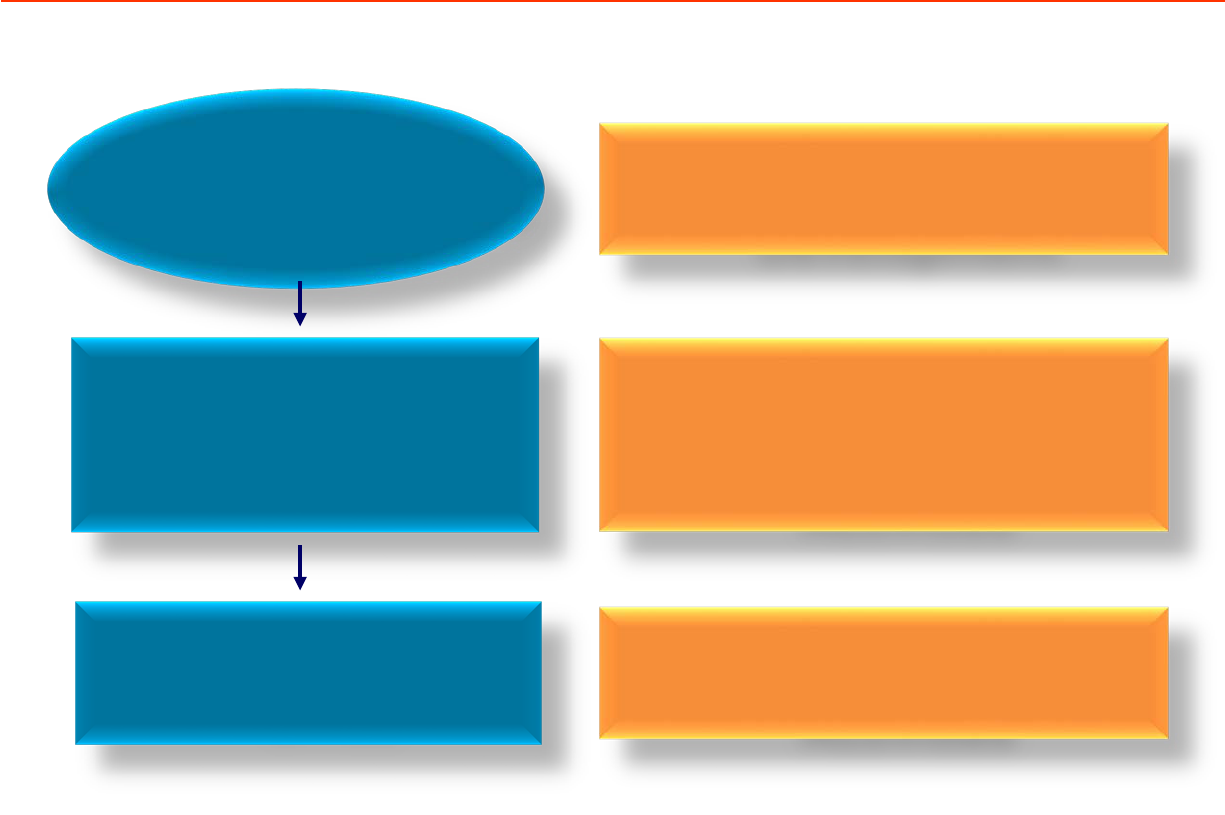
FSQD HEADQUARTERS
STATE HEALTH
DEPARTMENT
DISTRICT HEALTH
OFFICE
Planning and Policy
Development
Supervision on
Implementation of
Activities
Implementation of
Activities
INFRASTRUCTURE
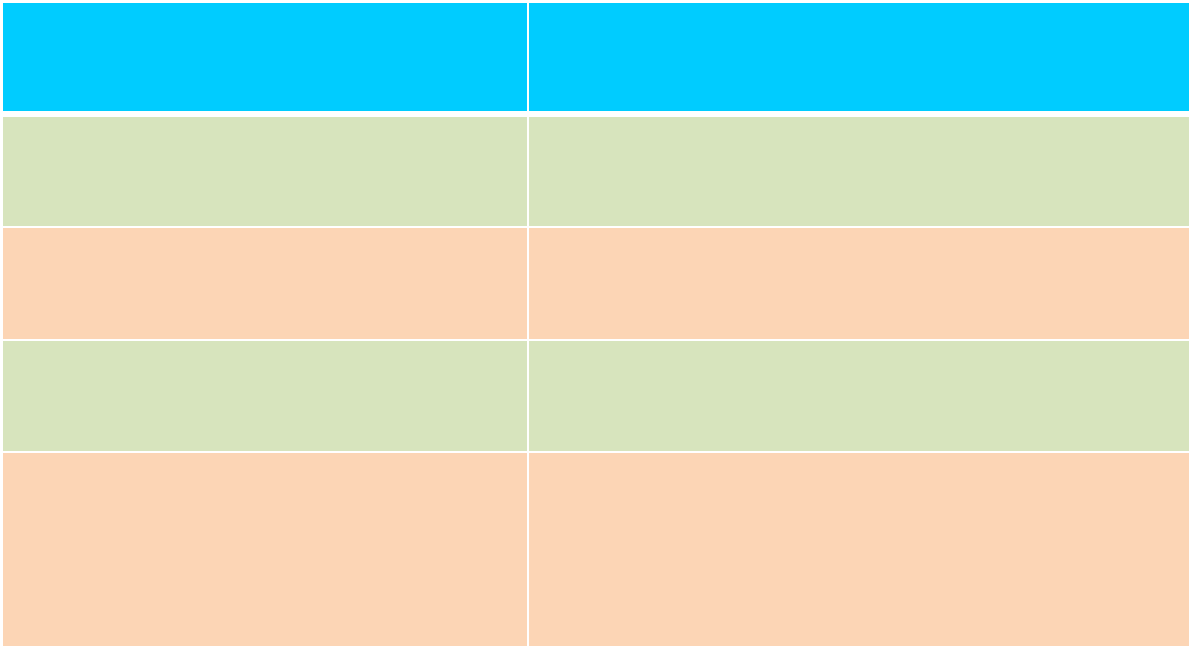
INFRASTRUCTURE NO.
STATES 15
DISTRICTS 136
ENTRY POINTS 56 (sea, land and air)
MOH LABORATORIES
15
(10 FSQLs and 5 PHLs)
Food Safety and Quality Programme
Ministry of Health Malaysia
7
FOOD LAWS
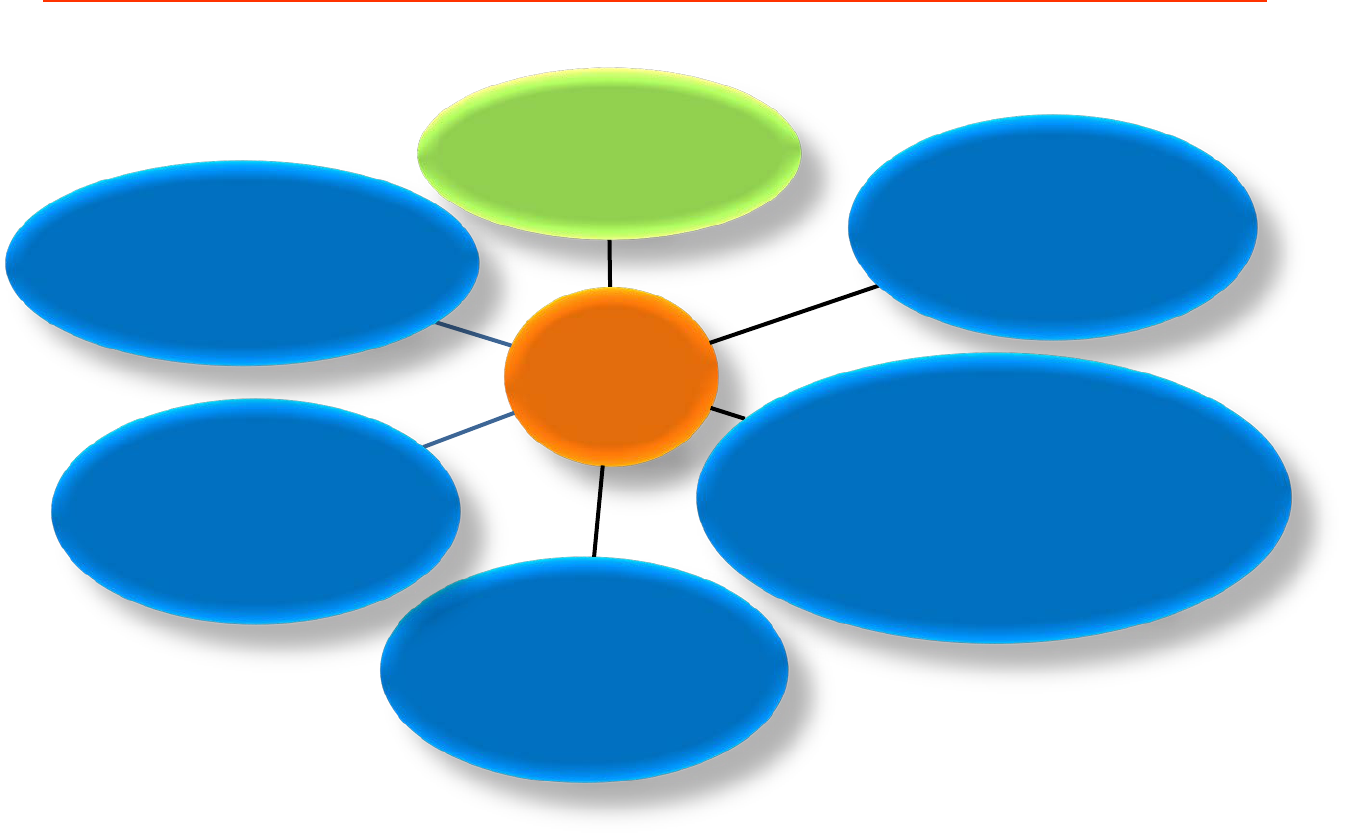
8
FOOD
ACT
1983
FOOD
REGULATIONS
1985
FOOD HYGIENE
REGULATIONS
2009
FOOD (ISSUANCE OF
HEALTH CERTIFICATE
FOR EXPORT OF FISH &
FISH PRODUCT TO THE
EUROPEAN UNION)
REGULATIONS 2009
FOOD
IRRADIATION
REGULATIONS
2011
FOOD (FOOD
ANALYSIS FEES)
REGULATIONS
2016
FOOD
(COMPOUNDING OF
OFFENCES)
REGULATIONS 2017
Food Safety and Quality Programme
Ministry of Health Malaysia

MRL Import activity are governed by 2 agencies as
below:
Food Safety & Quality Division (FSQD),
Ministry of Health, Malaysia (MOH)
Department of Agriculture (DOA),
Ministry of Agriculture and Agro-Based Industry
(MOA)
1
2
Food Safety and Quality Programme
Ministry of Health Malaysia

Roles and Authorities
Ministry of Health
Food safety
Quality Food stuffs
Disease identification
Testing materials include MRL
Inspections of Processors
Inspections of food establishments
Border inspections
Issuance of certificates for export & import
Department of Agriculture
Inspections
Border Control
Phytosanitary Certificates
Testing
Quarantine
Pesticide control
Farm Certification (SALM)
Companies
Set standards quality &
food safety
Supplier Contracts
Manufacturing
Advertising
Importing Countries
Inspections
Testing
Quarantine
In-economy inspections
Notifications
Destruction of materials
Import Standards
Consumers Union
Register complaints
Advocacy for consumers
Test samples for safety
Work with government to
improve overall performance
Food Chain
Processor
Exporter
Retailer
Restaurant
Kiosks
Importer
Consumer
Farm or
Fishing vessel
Feed
Water
Fertilizer
Pesticides
Intermediate
Processing
Ministry of Agriculture
Dept. of Veterinary Services
Inspections
Border Control
Sanitary Certificates
Testing
Vaccination, quarantine & disease free
zones
Animal Health Certificates
Ministry of Agriculture
Department of Fisheries
Inspections
Health Certificates
Aquaculture registration
Testing
Disease-free zones
Certification (SALM)
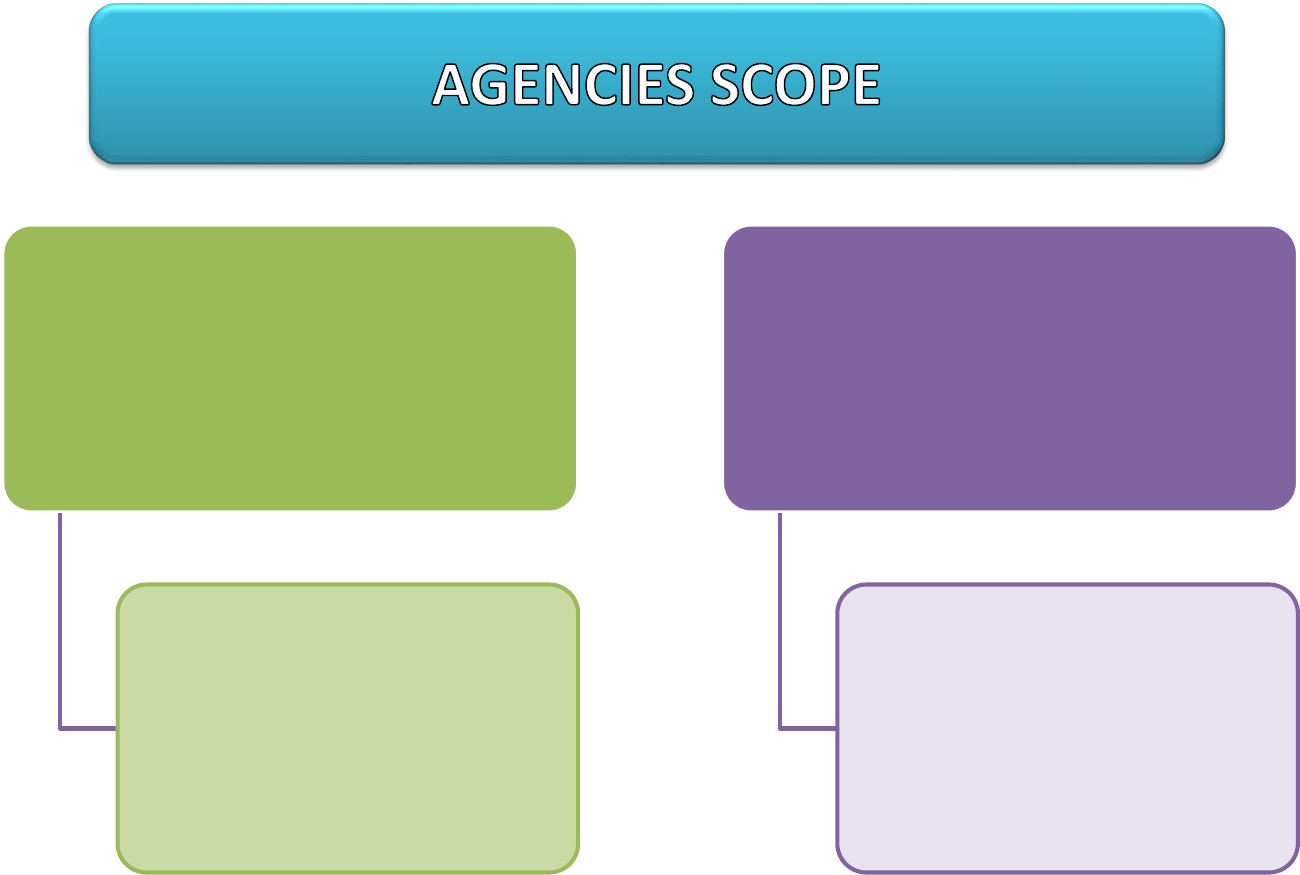
FSQD
Approve MRL and
enforcing MRL safety at
the point of sale
DOA
Register new Pesticide
and ensuring Good
Agriculture Practice (GAP)
at Farms
Food Safety and Quality Programme
Ministry of Health Malaysia


• Applicable for
Domestic and import
food product
• MRL stated;
a. Under regulation 41, 16
Schedule, The Food
Regulations 1985; or
b. Codex Alimentarius; or
c. 0.01 mg/kg
1. New MRL approval
• Submit application to
FSQD
• Up to 12 months process
to approve
2. Pesticide registration
• Submit application to
DOA
Food Safety and Quality Programme
Ministry of Health Malaysia

Food Safety and Quality Programme
Ministry of Health Malaysia

Food Safety and Quality Programme
Ministry of Health Malaysia

Food Safety and Quality Programme
Ministry of Health Malaysia
16
Notify the proposal of new regulation or amendment of current regulation to Malaysia Productivity Corporation
(MPC)
3 days
RIA IS NOT REQUIRED (PROCESS A)
RIA IS REQUIRED
1 month
STEP I
Proposed amendment/ issue will be discuss in Expert
Working Group
Conduct Regulatory Impact Analysis (RIA) and
consultation
2-6 month
1 month
STEP II
Food Regulation 1985
Advisory Committee deliberate the
proposal from expert working group
Submit Regulatory Impact Statement (RIS) to MPC.
Assesment and review of RIS by MPC
3 weeks
1-2 month
STEP III
Public Consultation (Online/ Dialog with stakeholdes /
WTO notification)
Submission of RIS to NDPC 1 week
1 month
STEP IV
Draft amendment will revised accordingly and translation
prepared
NDPC examines RIS for adequacy 3 weeks
1 month
STEP V
Draft amendment forwarded to Legal Advisor of Ministry of
Health for review
Provide RIS to decision maker. RIS is forwarded to the
Cabinet, Minister or other authority responsible.
2 month
STEP VI
Draft amendment forwarded to Attorney General Chamber
(AGC) for approval
Publication of RIS. MPC to consult regulator on publication
of RIS
PROCESS A
2 weeks STEP VII
Application of Minister of Health signature
STEP VIII
1 month New Regulation or amendment forwarded to AGC to be
AMENDMENT PROCESS
New MRL approval
12 Expert Working Group
under the Advisory
Committee on Food
Regulations 1985
Expert
Committee on
Nutrition/
Claims and
Advertisement
Expert
Committee
on Food
Additive
Expert
Committee on
Food
Contaminants
Expert
Committee on
Microbiology
Task Force on
Pesticide
Residues in
Food
Expert
Committee
on Drug
Residues in
Food
Ad-Hoc
Expert
Committee
on Water
Standard
Ad-hoc Expert
Committee on
Genetically
Modified Food
Expert
Committee
on Food
Labelling
Expert
Committee
on Food
Standard
Commodity
Expert
Committee
on Food
Packaging
Expert
Committee
on Food
Irradiation

Public Consultation
In line with the government's commitment to ensure that the
amendments are made in a transparent manner and takes
into account the views of relevant stakeholders
Online Public
Engagement
1 month
Dialogue with related
stakeholders, agencies and
consumer association
WTO Notification
60 days
Food Safety and Quality Programme
Ministry of Health Malaysia


• Domestic samples are taken from retail outlets in
domestic markets such as wet market,
supermarkets, hypermarkets, etc. while import
samples are taken at the entry points and send to
laboratory for analysis
• Pesticide residue screening and analysis in food
items is being conducted by accredited laboratory
under ISO 17025
• conviction, to a fine not exceeding one hundred
thousand ringgit (RM 100, 000.00) or to
imprisonment for a term not exceeding ten years or
both
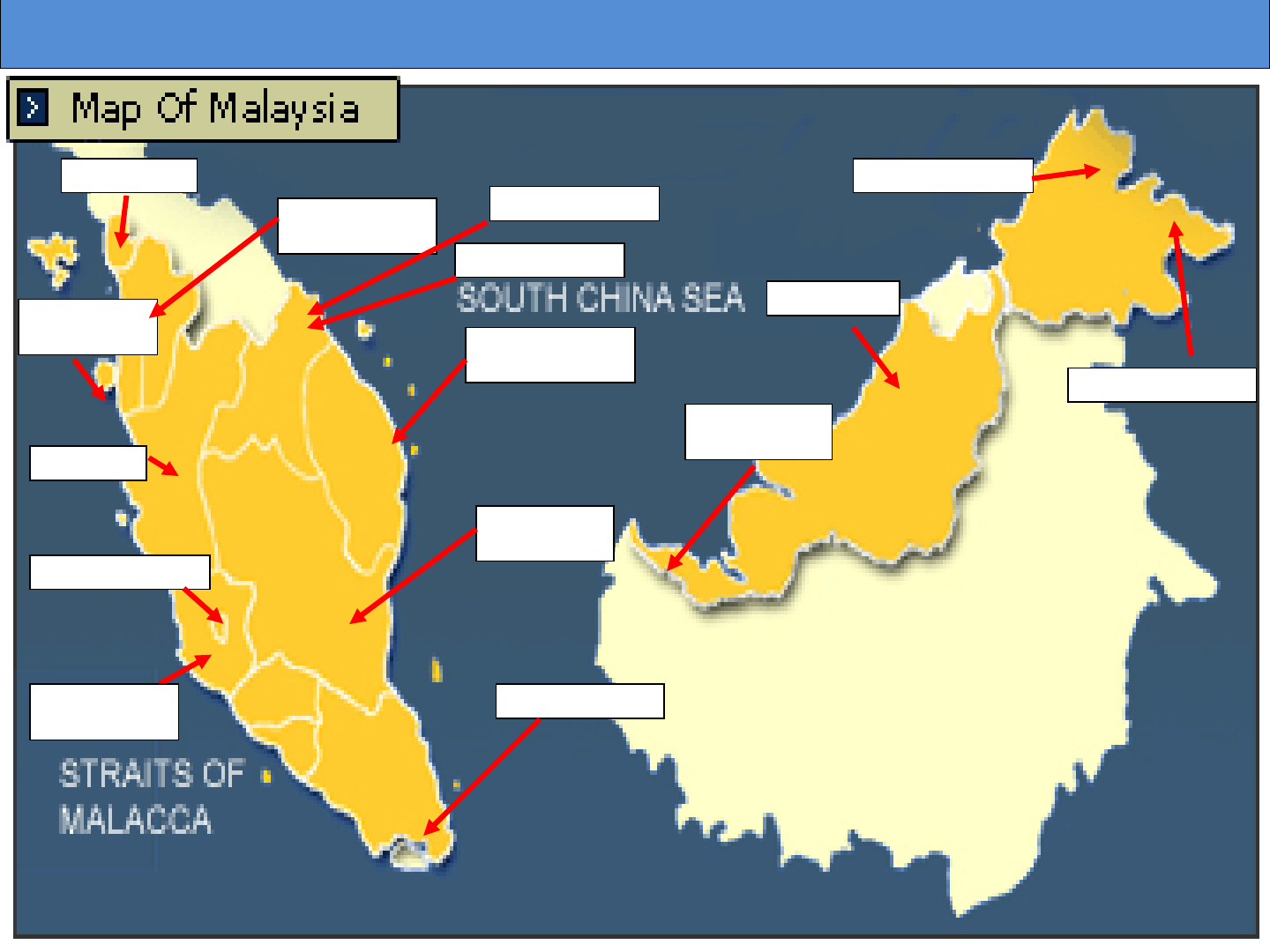
Food Safety and Quality Programme
Ministry of Health Malaysia
21
PHL Kota Kinabalu
FSQ LAB Miri
FSQ LAB Sandakan
FSQ LAB
Sarawak
FSQ LAB
Pahang
FSQ LAB
Terengganu
FSQ LAB Kelantan
PHL Johor Bharu
NPHL Sg Buloh
PHL Ipoh
FSQ LAB
Penang
FSQ LAB Perlis
FSQ LAB Alor
Setar
MOH Laboratories Throughout Malaysia
FSQ LAB
Selangor
PHL Kelantan

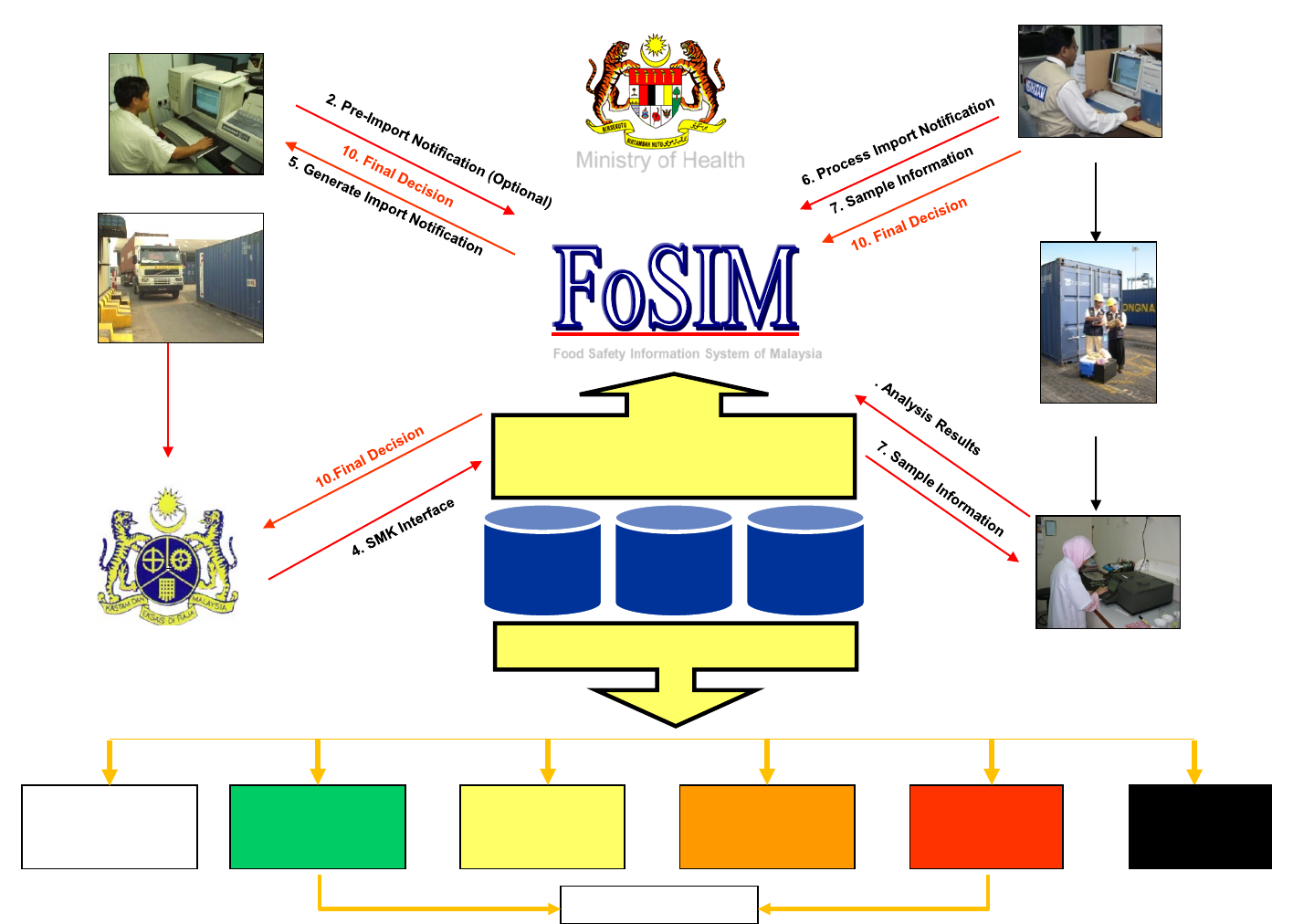
Transaction
Database
Master
Database
Knowledge
Database
LEVEL 1
AUTO
CLEARANCE
LEVEL 2
DOCUMENT
EXAMINATION
LEVEL 3
MONITORING
EXAMINATION
LEVEL 4
SURVEILLANCE
EXAMINATION
LEVEL 5
HOLD TEST &
RELEASE
LEVEL 6
AUTO
REJECTION
3. Import Declaration (K1)
IMPORTERS / FORWARDING AGENTS
FOOD SAFETY INFORMATION SYSTEM OF MALAYSIA – FOOD IMPORTATION PROCESS
8. Sample for Analysis
1. Registration
ENTRY POINT
INSPECTION / SAMPLING
LABORATORYCUSTOMS INFORMATION SYSTEM (SMK)
Food Safety Information System of Malaysia
Recommends
Examination Level
Automated Examination Level
Inspection Action
Ministry of Health
Food Safety and Quality Programme
Ministry of Health Malaysia
23

• Enforcement approach for MRL are coordinated effort
between multiple government agencies

25
Food Safety and Quality Programme
Ministry of Health Malaysia
Rafeah Sibil
Food Safety and Quality Division
Ministry of Health Malaysia
(rafeahsibil@moh.gov.my)
